Difference between revisions of "Absorption Coolers"
Jump to navigation
Jump to search
| (One intermediate revision by the same user not shown) | |||
| Line 7: | Line 7: | ||
* Please start editing this page after the /noinclude | * Please start editing this page after the /noinclude | ||
* -------------------------------------------------></noinclude> | * -------------------------------------------------></noinclude> | ||
[[File:ABSORPTION CHILLER.jpg|thumb|right|Absorption Coolers]] | |||
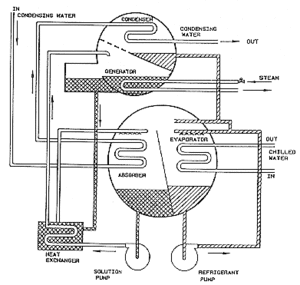
[[File:ABSORPTION CHILLER COMPONENTS.gif|thumb|right|Components of Absorption Coolers]] | |||
'''Absorption Coolers''' use heat rather than electricity as their energy source. Because natural gas is the most common heat source for absorption cooling, it is also referred to as gas-fired cooling. Other potential heat sources include propane, solar-heated water, or geothermal-heated water. | |||
Although mainly used in industrial or commercial settings, absorption coolers are commercially available for large residential homes. | |||
==An absorption cooling cycle relies on three basic principles== | |||
* When a liquid is heated it boils (vaporizes) and when a gas is cooled it condenses | |||
* Lowering the pressure above a liquid reduces its boiling point | |||
* Heat flows from warmer to cooler surfaces. | |||
Latest revision as of 13:32, 4 August 2012
Absorption Coolers use heat rather than electricity as their energy source. Because natural gas is the most common heat source for absorption cooling, it is also referred to as gas-fired cooling. Other potential heat sources include propane, solar-heated water, or geothermal-heated water. Although mainly used in industrial or commercial settings, absorption coolers are commercially available for large residential homes.
An absorption cooling cycle relies on three basic principles
- When a liquid is heated it boils (vaporizes) and when a gas is cooled it condenses
- Lowering the pressure above a liquid reduces its boiling point
- Heat flows from warmer to cooler surfaces.